Capillary action is a significant ability of water to move through other materials. Practical use of capillary action is visible in all kinds of our daily lives. The rise of a column of liquid within a narrow capillary tube is also because of the surface tension. Indeed, it often acts in opposition to gravity. This movement does not need the force of gravity to take place. It appears to have defied gravity by lifting up the straw. If you put a fine straw into a glass of water, You can observe that the water has mounted the straw and is more than the level of water in the glass. Capillary action definition is summed up as the impromptu flow of a liquid into a narrow tube or porous material. In 1905, Einstein published four seminal papers in the same journal these four papers are known as the Annus Mirabilis Papers.Capillary action is also sometimes called capillarity, capillary motion, or wicking. It was submitted in late 1900 and was published in 1901. It was titled Folgerungen aus den Capillaritätserscheinungen, which translates as Conclusions from the capillarity phenomena, found in volume 4, page 513. However, for a 2 cm wide (0.01 m radius) tube, the water would rise 1.4 mm, and for a 0.2 mm wide (0.0001 m radius) tube, the water would rise 140 mm (about 5.5 inches).Īlbert Einstein's first paper submitted to Annalen der Physik was on capillarity. Thus for a 2 m wide (1 m radius) tube, the water would rise an unnoticeable 0.014 mm. Differences in soil matric potential ( Ψ m. Capillary action is responsible for moving groundwater from wet areas of the soil to dry areas. In hydrology, capillary action describes the attraction of water molecules to soil particles. 
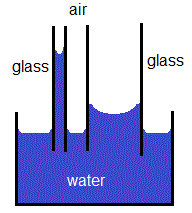
This flow can be calculated by Poiseuille's equation for compressible fluids. The term capillary flow is also used to describe the flow of carrier gas in a silica capillary column of a GC system. With some pairs of materials, such as mercury and glass, the interatomic forces within the liquid exceed those between the solid and the liquid, so a convex meniscus forms and capillary action works in reverse. For example, a glass capillary tube 0.5 mm in diameter will lift approximately a 2.8 mm column of water. The contact length (around the edge) between the liquid and the tube is proportional to the diameter of the tube, while the weight of the liquid column is proportional to the square of the tube's diameter, so a narrow tube will draw a liquid column higher than a wide tube. Surface tension pulls the liquid column up until there is a sufficient mass of liquid for gravitational forces to overcome the intermolecular forces. When the lower end of a vertical glass tube is placed in a liquid such as water, a concave meniscus forms. Some old sport and exercise fabrics use capillary action to "wick" sweat away from the skin.Ī common apparatus used to demonstrate capillary action is the capillary tube. Paper towels absorb liquid through capillary action, and capillary action is observed in the chemical separation technique known as thin layer chromatography. The same effect is what causes porous materials such as sponges to soak up liquids.Ĭapillary action helps move groundwater from wet areas of the soil to dry areas, and helps drain tear fluid from the eye. The effect causes a concave meniscus to form where the substance is touching a vertical surface. It occurs when the adhesive intermolecular forces between the liquid and a substance are stronger than the cohesive intermolecular forces inside the liquid. The standard reference is to a tube in plants but can be seen readily with porous paper. .jpg)
Capillary Flow Experiment to investigate capillary flows and phenomena aboard the International Space Station.Ĭapillary action, capillarity, capillary motion, or wicking is the ability of a substance to draw another substance into it. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
